Effect of kinase overexpression on growth and morphogenesis phenotypes
Since we found that the ubiquitin ligase SCFCDC4 functions as an inhibitor of hyphal morphogenesis, we set out to identify additional inhibitors of the yeast-to-mold switch. Identifying such inhibitors would improve our understanding of hyphal morphogenesis. We are carrying out screens to identify protein regulators (kinases and phosphatases) that inhibit hyphal formation when overexpressed. We are also screening collections of chemical compounds that inhibit hyphal morphogenesis, and are characterising their cellular targets. Both approaches point to specific cellular pathways that are differentially involved in mold vs. yeast growth.
a) Identification of kinases and phosphatases that inhibit hyphal morphogenesis
Since kinases are among the main regulators in all cells, in a collaboration with the Arkowitz and Morschhauser labs, we created a panel of all detectable Candida albicans genome open reading frames encoding kinases, phosphatases or their regulators (e.g. cyclins, kinase inhibitors), forcibly expressed under the TETon promoter. This panel was tested for growth under different stress conditions, as well as for induction or suppression of hyphal formation. Screening of this panel yielded several genes that consistently inhibit hyphal formation. We are focusing on one kinase in particular, which stunts the growth of hyphae when overexpressed, whereas deletion of this kinase results in accelerated hyphal elongation. This kinase, Akl1, regulates fluid-phase endocytosis, further corroborating the intimate link between endocytosis and morphogenesis. Another kinase regulator that suppresses hyphal morphogenesis is the G1 cyclin Cln3. With its CDK Cdc28, Cln3 has been known to affect endocytosis as well. We focus on another activity of Cln3, namely suppression of the activity of the central transcription factor of hyphal morphogenesis, Ume6.
b) Identification of chemical compounds that inhibit hyphal morphogenesis
Chemical compound libraries can be screened for chemicals that interfere with a cellular process, in order to gain understanding into its mechanism. In collaboration with the Kron group at the University of Chicago, we identified, from repurposing libraries, several compounds that inhibit hyphal morphogenesis, such as trifluoperazine, CGS 12066B and prochlorperazine.
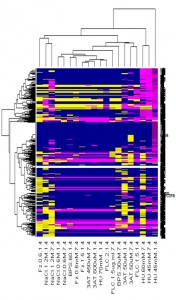
Taking advantage of the fact that some of these compounds are toxic in S. cerevisiae, we carried out genetic screens in this organism. We isolated genes that confer resistance to the identified compounds when overexpressed, and screened the complete yeast deletion collection for hypersensitive or -resistant strains in order to obtain clues to the cellular target(s) of these chemicals. Interestingly, most compounds isolated caused a strong reduction in fluid-phase endocytosis in S. cerevisiae as well as C. albicans, underscoring again the importance of this process in hyphal elongation.

Effect of cmpd 1 on an arrayed S. cerevisiae mutant collection (collaboration with Shay Ben-Aroya, Bar-Ilan U.)

