Like many fungal pathogens, Candida albicans is able to adopt multiple cellular morphologies. Under standard growth conditions it grows as individual yeast cells, whereas at high temperatures and in the presence of certain chemical signals, it adopts a filamentous, hyphal (mold) morphology. We found that mutants of the ubiquitin ligase SCFCDC4 grow constitutively as hyphae, indicating a role for ubiquitin-mediated protein degradation in the C. albicans morphogenetic switch. We identified the transcription factor Ume6 as the critical substrate of SCFCDC4 that is responsible for this mutant phenotype. Since SCFCDC4 substrates universally need to be phosphorylated in order to be recognized and degraded, we also investigated the role of kinases in this regulation. We found that the cyclin-dependent kinase Cdc28/Cdk1, in conjunction with the cyclin Hgc1, is necessary for degradation of Ume6. Interestingly, we find that in conjunction with another cyclin, Cln3, Cdc28/Cdk1 also down-regulates Ume6 function at the level of protein activity. Since Cln3 is essential for proliferation in the yeast form, its effect on Ume6 activity might explain the antagonism between yeast proliferation and hyphal differentiation in Candida albicans.
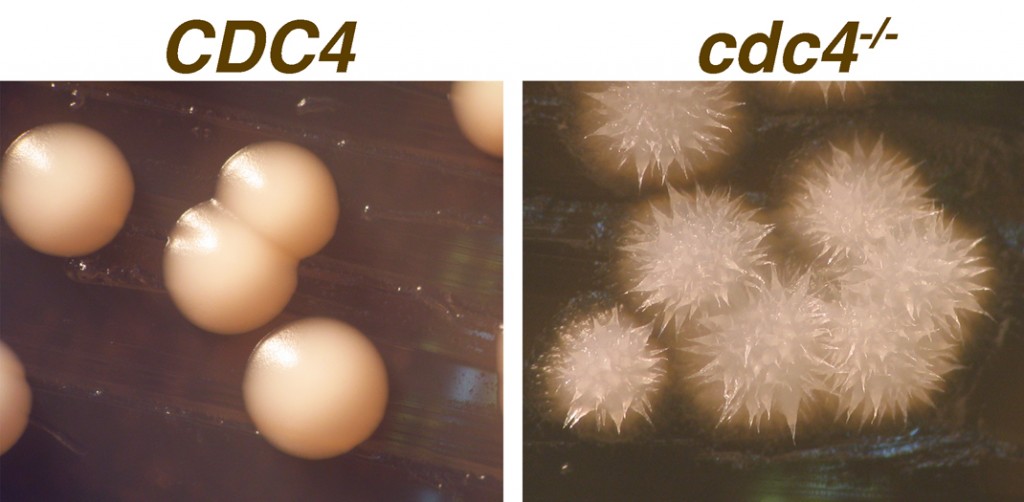
C. albicans cdc4-/- strains make highly unusual colonies, shown on the right in the image below.
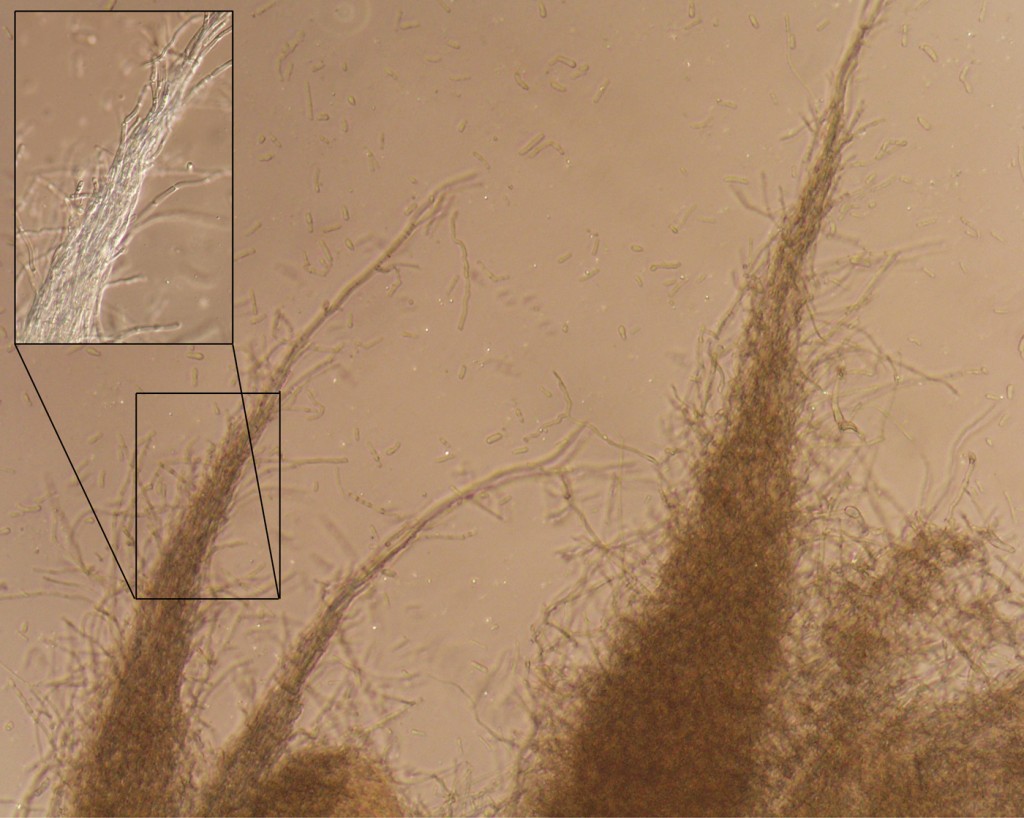
The hairy protrusions are made of bundles of extended hyphae as shown in the picture below.
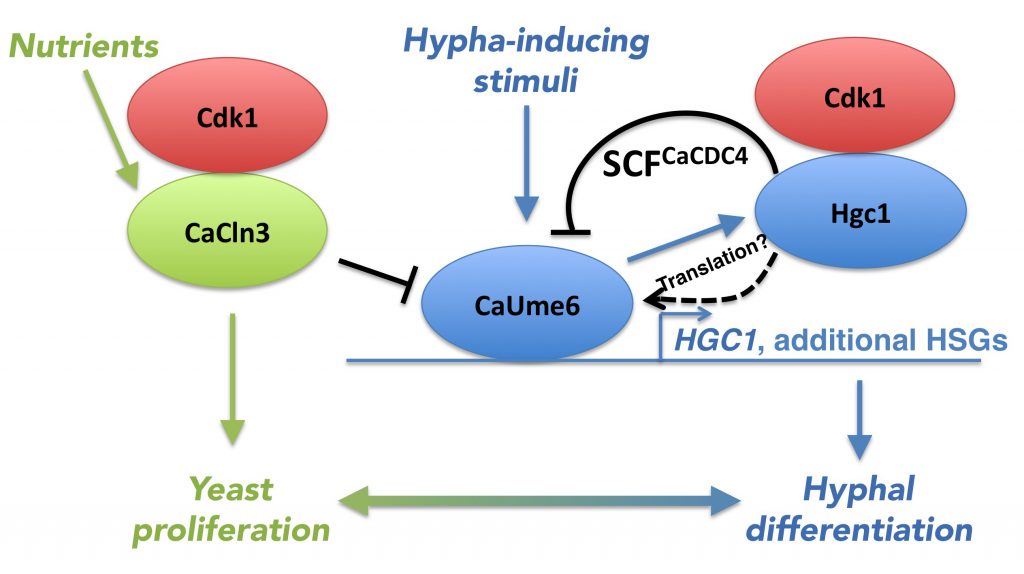
Right hand side, blue color: CaUme6 induces HGC1 alongside additional hypha-specific genes (HSGs), and Hgc1 together with Cdk1 induces SCFCaCDC4-mediated degradation of CaUme6. Left hand side, green color: the inducer of proliferation CaCln3 is activated by nutrients and represses the activity of CaUme6 and thus hypha formation. Black: negative regulations of CaUme6.
The CDK1 inhibitor Sol1
One interesting substrate of SCFCDC4 that we identified, the cyclin-dependent kinase inhibitor Sol1, is only peripherally involved in hyphal morphogenesis. However it is centrally involved in the coordination of cell growth and proliferation, as can be seen in the time-lapse movies below.
Wild-type or sol1-/- cells from an overnight culture were inoculated on an agar pad and photographed by time-lapse microscopy at 1 frame/min. Note how some of the sol1-/- cells stop proliferating but continue to increase in volume, until they explode.
Wild-type cells
sol1-/-
sol1-/-